SOLVED: 7 The heat of vaporization of freon (CCI Fz) is 17.2 kJlmol at 25 *C. What is the change of entropy for one mole of liquid freon when it vaporizes at

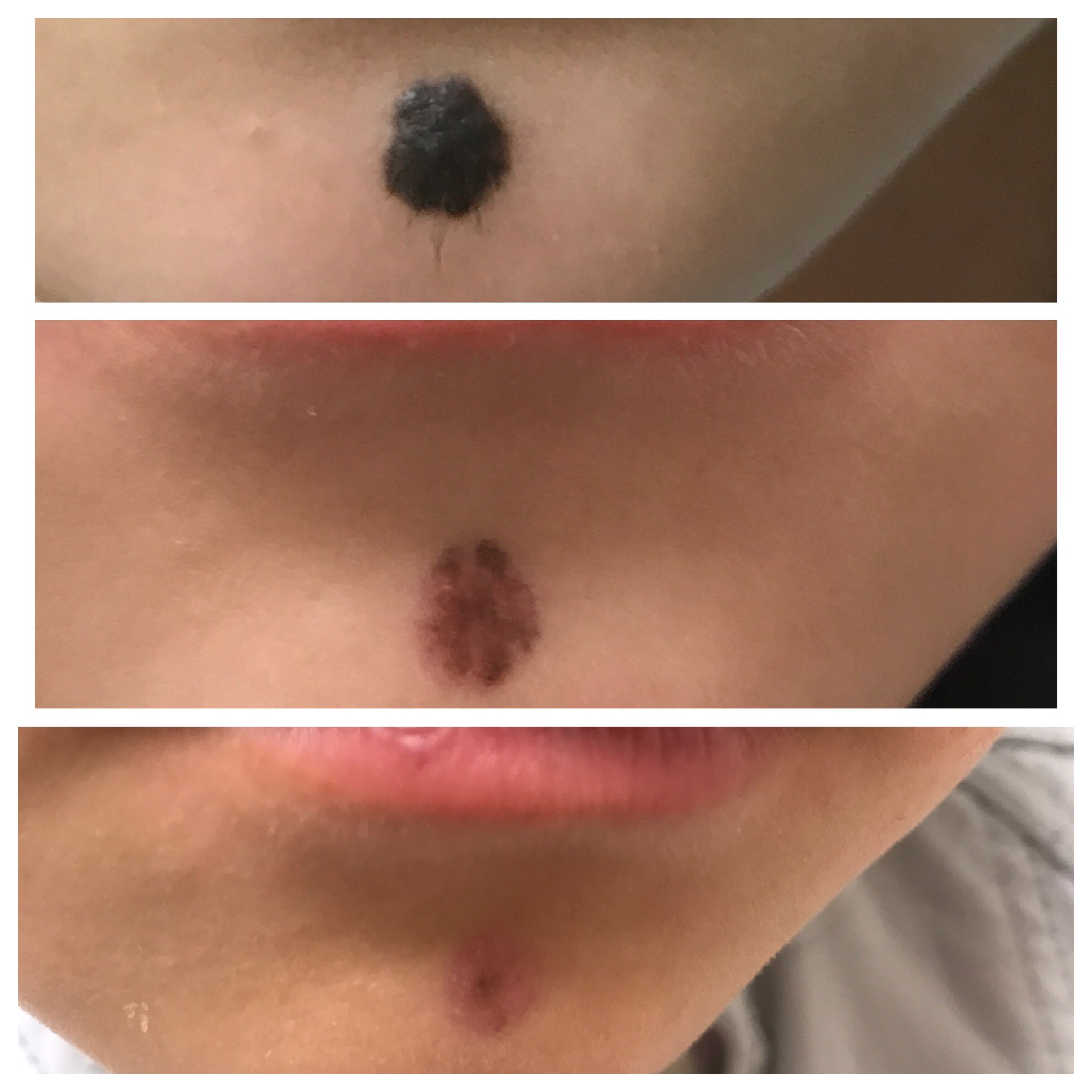
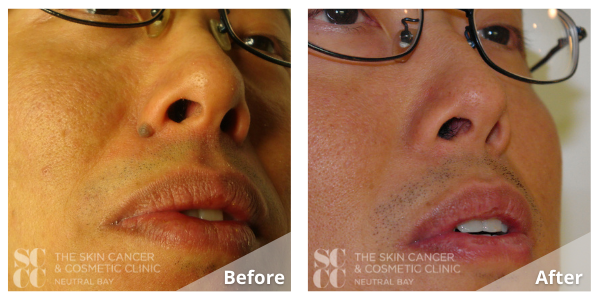
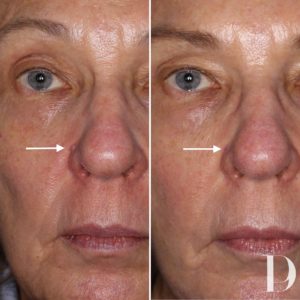
Live Laser Mole Removal. See Dr Young Vaporize a Mole with this Live Demo. Learn About Mole Removal - YouTube

CHEM 201 - Finding mole fraction from vapor pressure of a mixture with two volatile liquids - YouTube

The Clinic Ave Pampanga - Mole Removal before and right after laser vaporization procedure. Call or text us at 09173038027 for appt or PM us here on FB. :) #TheClinicAve #MedicalAesthetics #Pampanga #
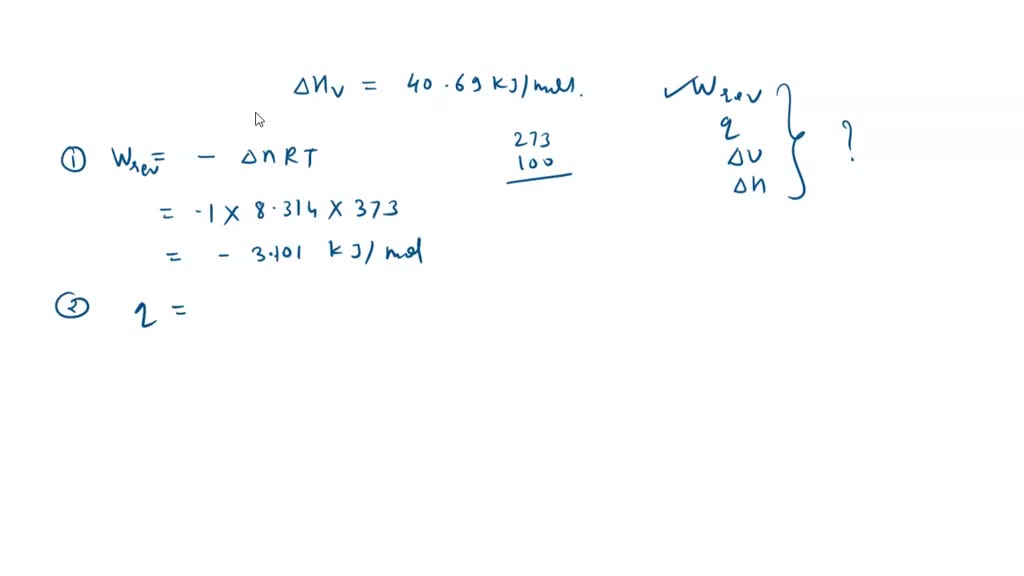
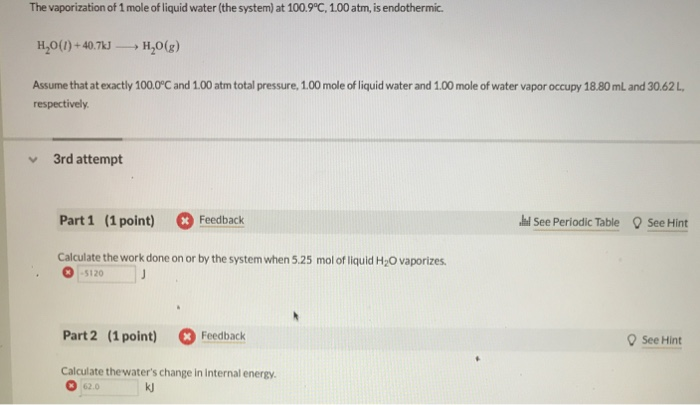
SOLVED: One mole of liquid water is vaporized at 100.0°C and 1.013 bar. The heat of vaporization of water at this temperature is 40.69 kJ/mol. What are the values of: w, q,
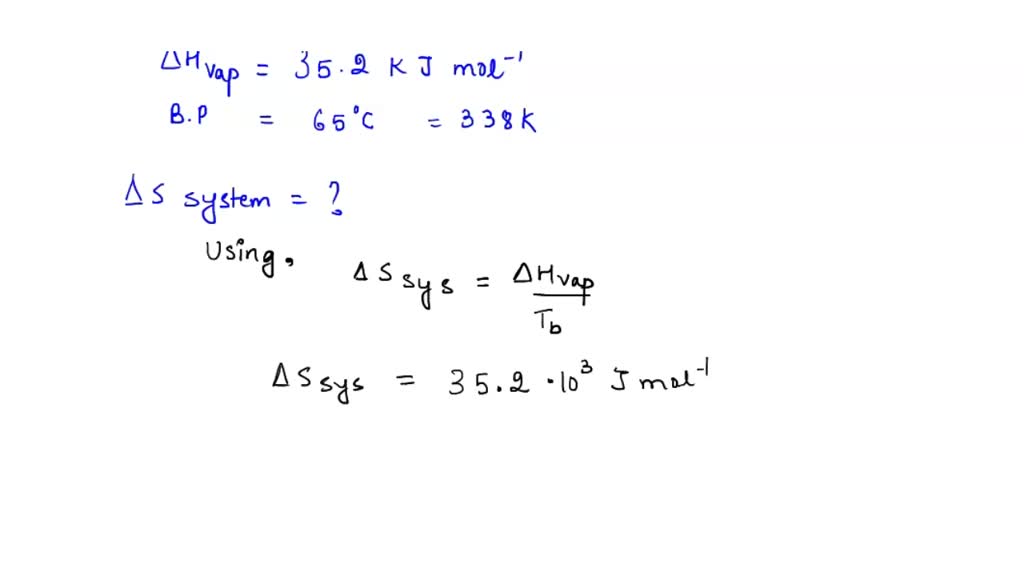
SOLVED: The enthalpy of vaporization of methanol is 35.2 kJ/mol at its boiling point (65°C). Determine ΔSsys, ΔSsurr, and ΔSuniv when 1.00 mole of methanol is vaporized at 65°C and 1.00 atm.






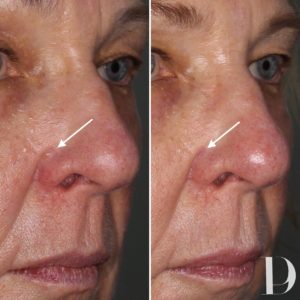

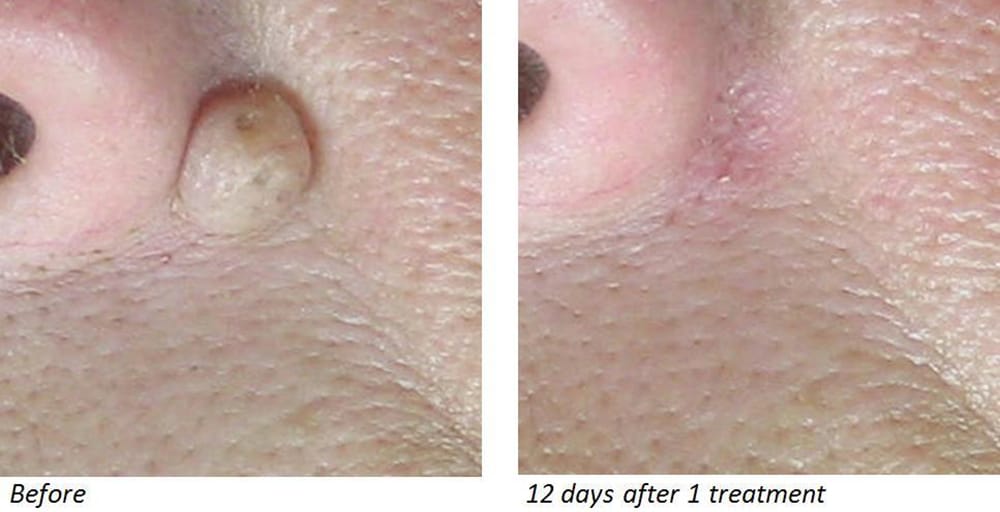
![The Clinics That Do the Best Mole Removal in Calgary [2023 ] The Clinics That Do the Best Mole Removal in Calgary [2023 ]](https://www.thebestcalgary.com/wp-content/uploads/2020/12/Best-Mole-Removal-in-Calgary-1-1024x576.jpg)